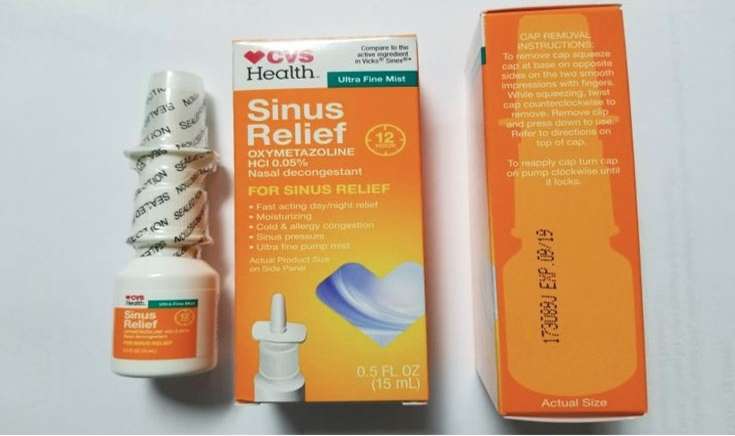
(WSVN) - CVS is recalling one of its private-brand nasal sprays due to a microbiological contamination.
Product Quest Manufacturing announced Thursday the recall of CVS Health 12 Hour Sinus Relief Nasal Mist Thursday, after it was determined that the product was contaminated with Pseudomonas aeruginosa.
Repetitive use of the nasal decongestant spray could lead to an infection that can be life-threatening for people with weakened immune systems, the FDA said.
The company has not received any reports of adverse events related to this recall.
The 16,896 units affected by the recall have a UPC code of 50428432365 and Lot # 173089J printed on them, with an expiration date of September 2019. The FDA says the sprays were distributed to CVS stores nationwide.
Product Quest is notifying its customers by oral and written communication and is arranging for return or replacement of all recalled products.
Consumers with questions can contact the company at (386) 239-8787, Monday through Friday from 8 a.m. to 4 p.m.
Copyright 2024 Sunbeam Television Corp. All rights reserved. This material may not be published, broadcast, rewritten or redistributed.